
Scientists Brought a 3-Billion-Year-Old Enzyme Back to Life, and It Still Works!
Nitrogen is everywhere in Earth’s atmosphere, yet virtually no organism can use it directly. That job falls to enzymes called nitrogenases, which convert atmospheric nitrogen into forms that plants, animals, and microbes can actually absorb. Understanding how those enzymes worked billions of years ago has long been one of science’s more stubborn puzzles, until now.
The study, published in Nature Communications, was conducted by researchers with the NASA-funded Metal Utilization and Selection across Eons (MUSE) project, a collaboration between Utah State University and the University of Wisconsin-Madison. Rather than relying solely on fossilized rocks, the team used synthetic biology to rebuild ancient nitrogenase genes from scratch and insert them into living bacteria, a first-of-its-kind experimental approach that transforms molecular paleontology from inference into direct observation.
Resurrecting Ancient Enzymes in the Lab
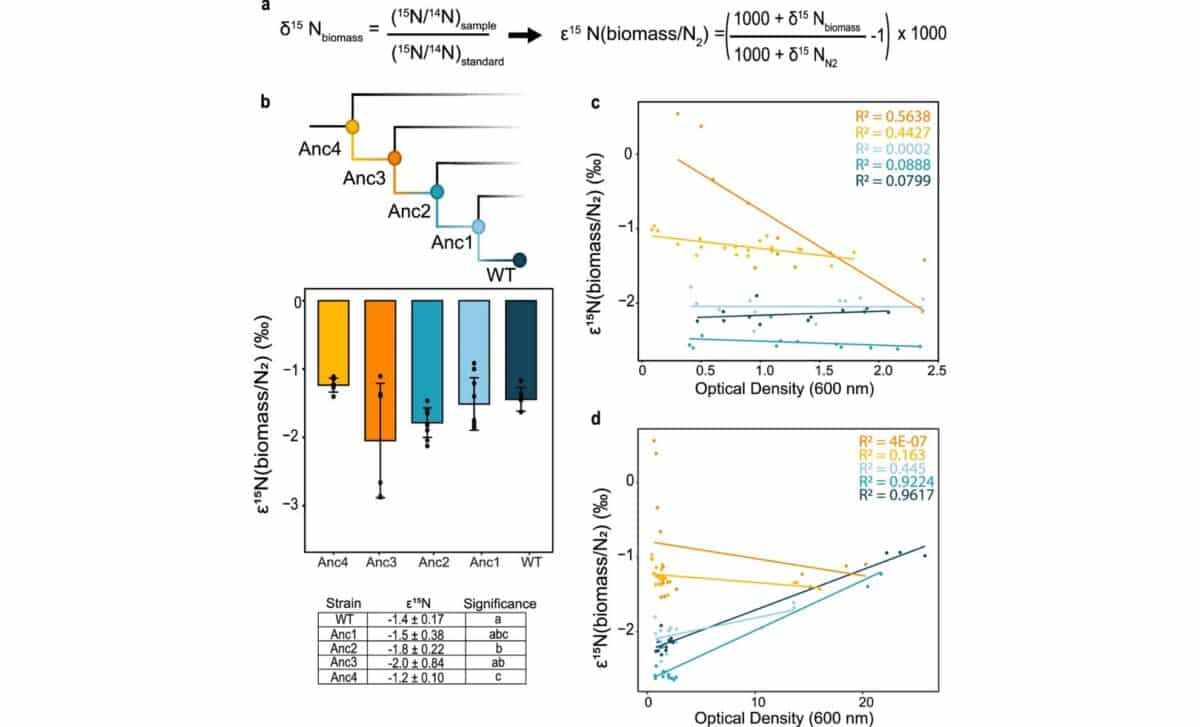
The research team constructed a library of four ancestral nitrogenase variants, labeled Anc1 through Anc4, representing a temporal sequence stretching from roughly 700 million to 2.3 billion years ago. According to the study, these variants were reconstructed from phylogenetic nodes along an oxygen-tolerant lineage of nitrogenase evolution and were engineered into a nitrogen-fixing bacterium called Azotobacter vinelandii, replacing its native nitrogenase genes entirely.
Utah State University biochemist Lance Seefeldt, who has spent more than 30 years studying nitrogenase structure and function, described the significance of the approach. “Until now, science has relied on ancient rock and fossils to study early life,” he said. “Study of fossilized enzymes assumes ancient enzymes produced the same isotopic signatures as modern enzymes.” The new framework tests that assumption directly rather than taking it on faith.
According to the paper, all four ancestral strains successfully supported diazotrophic growth, meaning they could fix nitrogen and sustain life, though they showed lower catalytic rates than modern wild-type enzymes. The oldest reconstructed ancestor, Anc4, was phylogenetically the most distant from its modern counterpart, yet it still functioned.
A Biosignature Frozen in Deep Time
The central finding concerns nitrogen isotope fractionation, the subtle way nitrogenase enzymes discriminate between heavier and lighter nitrogen atoms during fixation, leaving a chemical fingerprint in biological matter. Geologists have long used these signatures, preserved in ancient sedimentary rocks, as evidence for biological activity billions of years ago. The validity of that approach, however, depended on an untested assumption: that ancient nitrogenases left the same fingerprints as modern ones.
According to the Nature Communications study, all four ancestral enzyme variants produced isotopic fractionation values (ε¹⁵N) ranging from −0.9 to −2.9‰, a narrow window that directly overlaps with both modern nitrogen-fixing microbes and the isotopic signatures preserved in Archean sedimentary rocks dating back 3.2 billion years. The average fractionation across all ancestral strains was −1.6 ± 0.5‰, strikingly consistent across more than two billion years of molecular evolution.
Betül Kaçar, professor of bacteriology at the University of Wisconsin-Madison and the study’s corresponding author, framed the broader stakes plainly. “The search for life starts here at home, and our home is four billion years old,” she said. “We need to understand life before us, if we want to understand life ahead of us and life elsewhere.”
What This Means for Earth’s History, and Beyond
The isotopic consistency carries an unexpected implication: the origin of molybdenum-dependent nitrogenase may be even older than the currently accepted estimate of 3.2 billion years. According to the study, the similarity between the lowest nitrogen isotope values in early Archean rocks and the fractionation signatures produced by the reconstructed ancestral enzymes suggests the currently accepted minimum age “may be too conservative.” The researchers cite recent evidence for elevated dissolved molybdenum concentrations in Archean seawater as additional support for an earlier emergence.
The findings also push back against a competing hypothesis, that lower oxygen levels in the ancient Archean atmosphere enhanced isotopic fractionation through gas diffusion. According to the paper, the stability of ancestral nitrogenase-catalyzed fractionation across periods of widely fluctuating oxygen concentrations argues that environmental oxygen did not significantly influence the enzyme’s isotopic discrimination. The biochemical mechanism, it appears, was established early and held firm.
Beyond Earth history, Seefeldt noted that understanding both ancient and modern nitrogenases carries practical significance. “Understanding nitrogenases, both ancient and modern, is critical to helping us tackle current agricultural challenges in a changing climate, including areas at risk of famine due to drought and lack of access to commercial fertilizers,” he said. The same enzyme at the center of a two-billion-year-old mystery may also hold keys to feeding people in an uncertain future.
First Appeared on
Source link






