
A Flesh-Eating Fungus Spreading from Cats to Humans Has Been Detected, and It’s Expanding Fast
In veterinary offices along the southeastern coast of Uruguay, the cases did not arrive all at once. One cat came in with raw sores around the nose and paws. Another had crusted wounds across the face that were not healing as expected. Then another showed the same kind of damage, again centered on the head and forelimbs, again lingering longer than a routine infection should.
Not far away, clinicians were seeing patients with their own puzzling marks. Small bumps appeared on hands or forearms after scratches. Some lesions opened. Others multiplied and seemed to move beneath the skin rather than stay confined to one place. The pattern was specific enough to be memorable, but not yet simple enough to explain at a glance.
The reports stretched across Maldonado and Rocha, two departments on the country’s southeastern edge. There was no single household, no one animal, no obvious starting point linking all of them together. What stood out instead was repetition: similar wounds, similar contacts, similar timing, and a growing sense that the same story was surfacing in different rooms.
The Cases Kept Pointing Back to Cats
The picture became clearer through work associated with Universidad de la República, Uruguay’s main public university, where researchers documented infections in people, pets, and other local animals and tied the pattern to Sporothrix brasiliensis. The university’s site situates the institution at the center of the country’s research and public health training network, giving the investigation a clear national base.
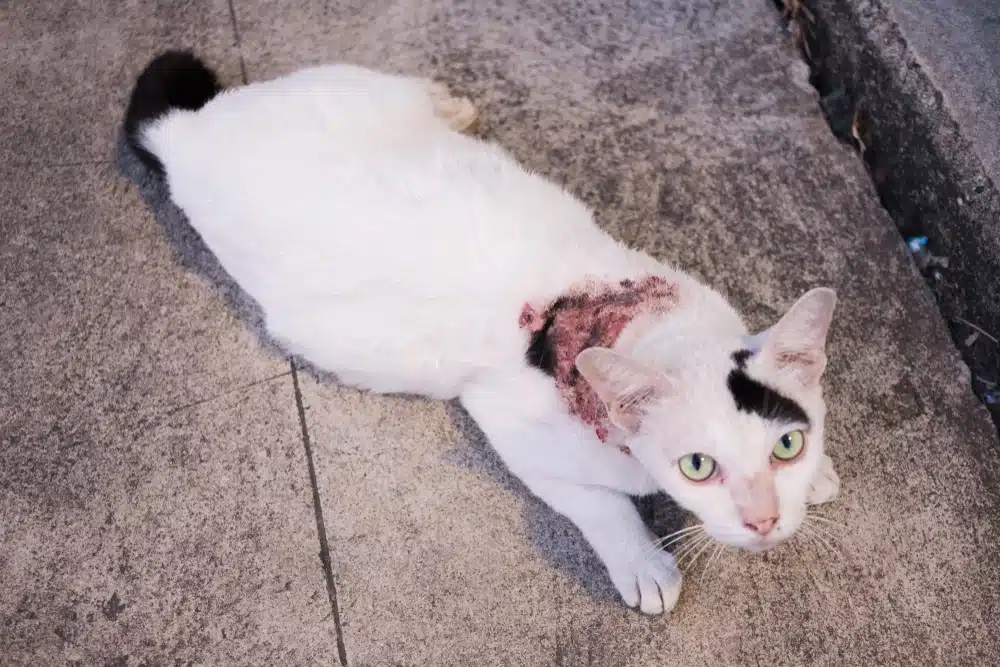
Cats matter here for a practical reason. Open wounds on infected cats can carry large amounts of the organism, especially around the mouth, face, and paws. When claws, teeth, or wound fluid break human skin, the fungus gains a direct route into tissue, making animal-to-person spread far more efficient than a casual description of a skin infection might suggest.
That helps explain why the early cases did not look like a random collection of isolated complaints. They shared the same visible features, the same animal contact, and the same stubborn refusal to resolve quickly. By the time those details lined up, the scene had shifted from a string of strange wounds to a recognizable clinical pattern.
A Fungus That Changes Shape With Heat
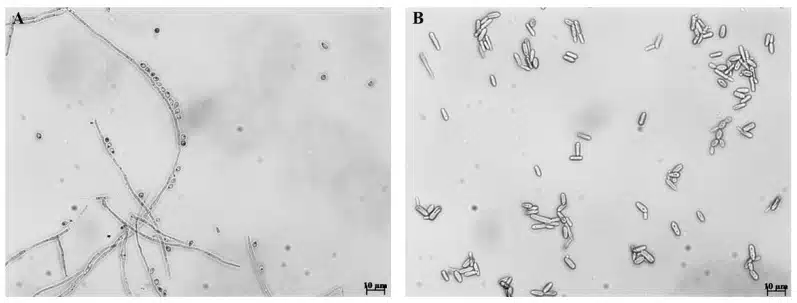
A 2023 review ppublished in the Journal of Fungi describes sporotrichosis as a disease caused by fungi in the genus Sporothrix and identifies S. brasiliensis as the “most virulent species” within the pathogenic clade. The phrase appears in the abstract itself, making it one of the clearest source-backed descriptions of why this organism has drawn so much attention across Latin America.
One reason it is so effective is that it is a thermodimorphic fungus. Outside the body it grows as branching filaments. Inside a mammalian host it converts to a yeast-like form better suited to surviving in tissue. The review links that shift, along with other virulence-related traits, to the organism’s ability to move between environment and host.
That biology matches what clinicians were seeing on the ground. In people, skin lesions often begin as a bump that later ulcerates, followed by nodules that track along the lymphatic system. In cats, the disease can look harsher, with persistent wounds, crusts, hair loss, and inflamed eyes. What seemed at first like separate complaints in separate species turned out to follow the same medical logic.
Uruguay’s Older Pattern Did Not Look Like This
Uruguay had seen sporotrichosis before, but not mainly in this form. A national study, available through PubMed by related review context and discussed in the regional literature, points back to a long history in which many diagnosed cases were linked to armadillo scratches during hunting rather than domestic animals moving through neighborhoods.
That older pattern mattered because it was narrower. Exposure stayed tied to a specific activity, in a specific setting, among a more limited group of people. It did not depend on cats crossing between homes, streets, and shelters, or on repeated contact between household animals and humans in everyday life.
The newer pattern is harder to map. A disease associated with hunting can remain sporadic for years. A fungus moving through cats behaves differently, because cats fight, roam, groom, and carry infection across the same spaces where people live. That is what turns a familiar diagnosis into a more complicated zoonotic transmission problem.
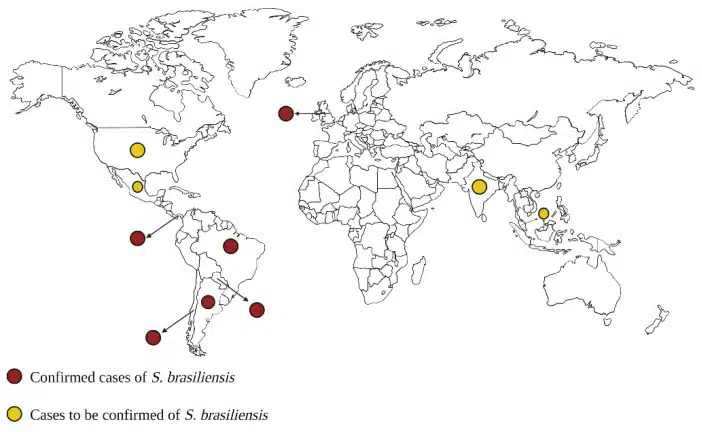
The Regional Trail Now Runs Through Argentina Too
The wider picture appears in a 2024 case report on ScienceDirect. The abstract describes S. brasiliensis as a “highly virulent emerging pathogen” and reports two human cases in Buenos Aires after contact with sick cats. It also says genetic analysis suggested the Argentine isolates were introduced from Brazil and likely came from the same source.
That regional evidence does not prove every Uruguayan case followed the same route. It does show, however, that cat-associated spread is not confined to one country. The same organism has already been documented moving through neighboring settings in ways consistent with animal-linked infection, which gives Uruguay’s cases a broader frame without overstating what has been established locally.
What remains, once the lesions and locations are stripped down to essentials, is a public health challenge that starts with recognition. The marks first seen in cats and then in people were not important because they were dramatic. They mattered because they repeated, across places and species, until they formed a pattern that could no longer be dismissed as incidental.
First Appeared on
Source link






