A Deadly Fungus That “Eats Human Body from Within” Is Spreading Rapidly Across the Planet
The global distribution of the Aspergillus fungus is undergoing a significant geographic reallocation. Researchers have identified a measurable northward migration of these airborne pathogens into regions previously considered too cold for their survival. This shift brings a higher concentration of infectious spores into densely populated areas of Northern Europe and North America.
The movement is not a random dispersal but a structured response to rising annual mean temperatures. As traditional habitats in the Southern Hemisphere exceed the thermal limits for fungal growth, the pathogens are finding new environmental niches. This expansion is happening quietly in the soil and the air, often undetected until the fungus enters clinical settings.
The public health implications are centered on the concept of environmental resistance. In the wild, Aspergillus is exposed to the same class of antifungal chemicals used in human medicine. This overlap creates a pre-selection process where only the most resilient strains survive to be inhaled by vulnerable individuals.
The University of Manchester led a multi-institutional study to map this progression through 2025. On May 2025, the research team released geospatial data confirming that the climate-driven migration of Aspergillus exposes an additional 9 million European residents to these spores. The findings, published in a Research Square preprint, utilize soil metabarcoding to track the pathogen across the continent.
A Continent Under Spore Pressure
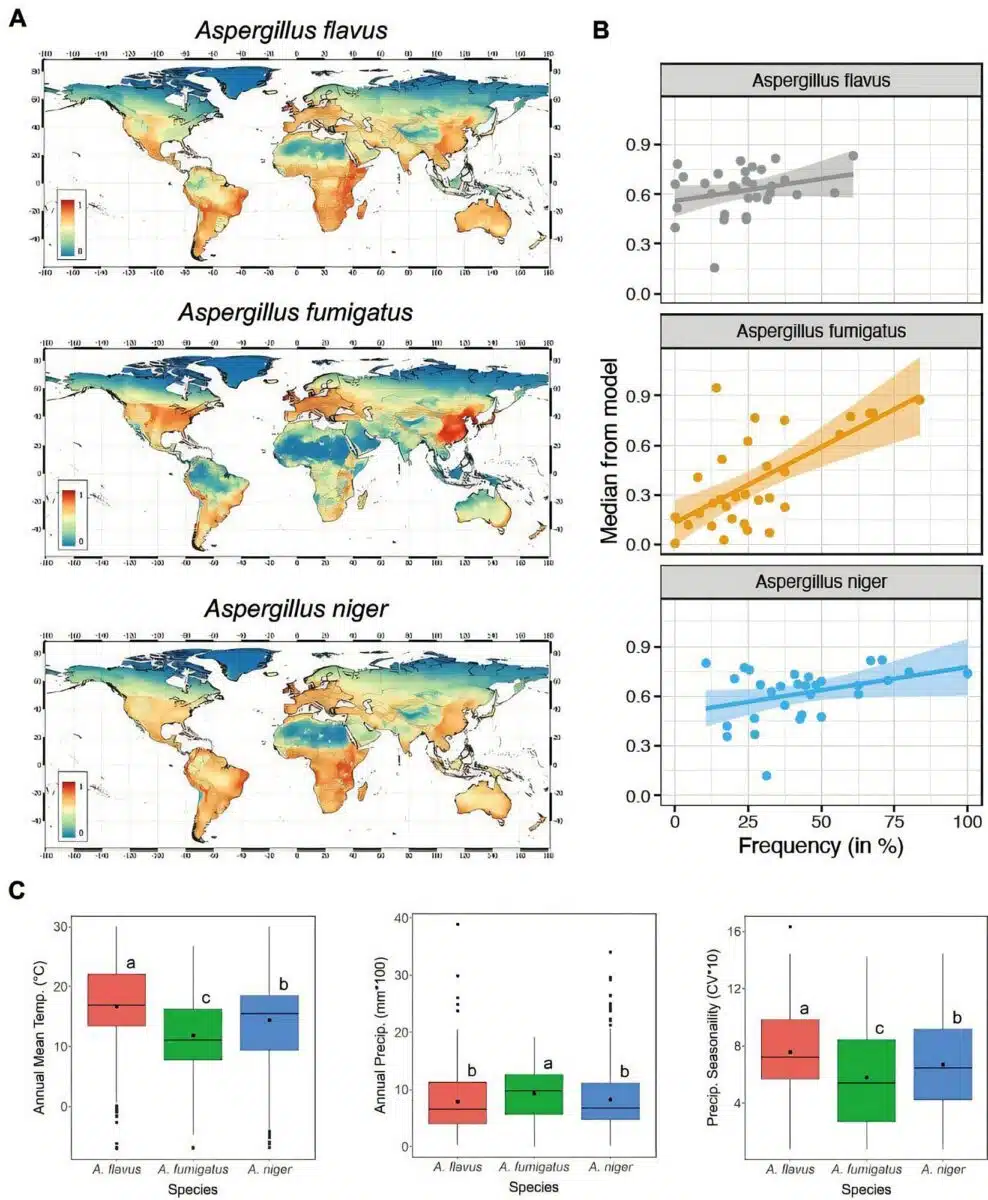
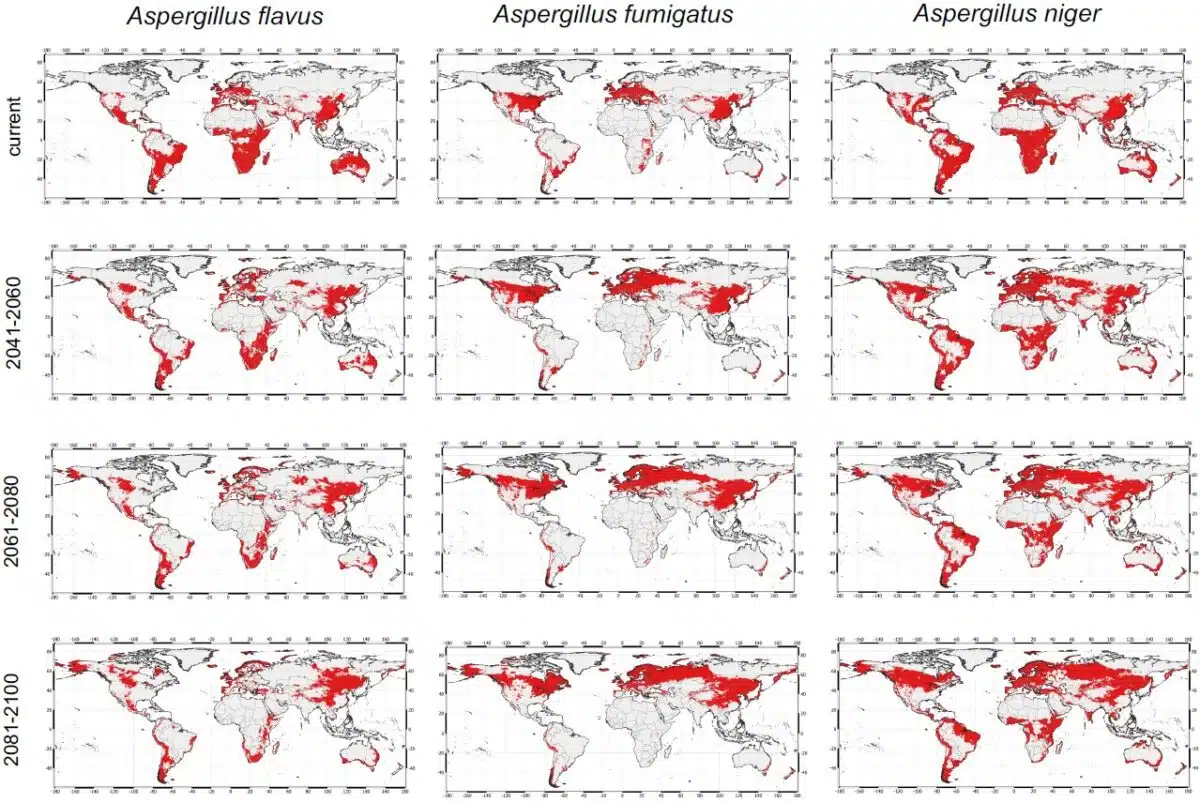
The data tracks three primary species: Aspergillus fumigatus, Aspergillus flavus, and Aspergillus niger. Under high-emission climate trajectories, the geographic footprint of A. fumigatus is projected to expand by 77.5 percent across Europe. Currently, 1.98 billion people worldwide live in environments suitable for this fungus. It is the primary cause of invasive aspergillosis, a condition with high mortality rates for immunocompromised patients.
The Manchester study indicates that warming zones are displacing these pathogens from tropical regions. The temperature shift concentrates spores in northern latitudes. Scandinavia and Alaska are specifically identified as new exposure centers. While parts of Africa and South America are becoming too hot for A. flavus, the fungus is expanding into Russia and northern China.
The research was coordinated with the Liverpool School of Tropical Medicine and the UK Centre for Ecology & Hydrology. The Wellcome Trust provided the financial support for the project. The modeling utilizes the SSP585 warming scenario, which assumes a continued global dependence on fossil fuels through the end of the century.
Under this framework, the baseline habitat for A. flavus in Europe increases by 16 percent. This expansion places an additional one million individuals within the exposure zone. Currently, 905 million people reside in zones suitable for A. niger, while 846 million live in areas supporting A. flavus.
From Farm Furrows to Hospital Wards
The environmental restructuring of fungal habitats directly impacts global food systems. Aspergillus species frequently infect maize and rice crops. This contamination spoils yields and introduces toxic aflatoxins into the food supply. In the United States, Aspergillus contamination results in annual losses of up to $1$ billion for the corn industry.
The expanded environmental footprint overlaps with industrial agricultural zones. Farmers utilize azole-based fungicides to protect crops from seasonal rot. Because medical professionals rely on nearly identical azole compounds to treat human infections, the fungus develops cross-resistance in the environment.
Dr. Norman van Rhijn directed the mapping project at the University of Manchester. He stated that “changes in environmental factors, such as humidity and extreme weather events, will change habitats and drive fungal adaptation and spread.” He noted that these organisms remain “relatively under-researched compared to viruses and parasites,” despite their growing global reach.
Viv Goosens, Research Manager at Wellcome, reviewed the data. She stated, “Fungal pathogens pose a serious threat to human health by causing infections and disrupting food systems. Climate change will make these risks worse.”
Mapping the Microscopic Threat
The predictive framework uses global DNA sequencing data from soil samples. The research team processed this data through a Maximum Entropy algorithm. The researchers combined these environmental datasets with a 1-kilometer resolution human population database and the CROPGRIDS global crop distribution index.
The model limits its variables to temperature, precipitation, and land-use metrics. It establishes thresholds for suitable habitats using the Maximum Test Sensitivity Plus Specificity method. Researchers verified the accuracy of the model through Receiver Operating Characteristic curves.
The methodology identifies annual mean temperature as the most influential metric for fungal habitat suitability. Aspergillus spores measure between two and three micrometers. This size allows them to bypass respiratory defenses and enter the pulmonary alveoli. The spores release from environmental reservoirs such as agricultural compost heaps. These heaps are hotspots because their internal temperatures often exceed 50 degrees Celsius.
Clinical incidence rates generally track with these distribution maps. In 14 national cohorts measured during the study, countries with a high environmental presence of a specific strain reported higher clinical rates of infection. This correlation confirms that environmental density drives hospital admissions.
The clinical response to these infections is often delayed. Physicians must use complex diagnostic tools including computed tomography scans and polymerase chain reaction analysis. These tests take time to process. The delay allows antifungal resistance strains to proliferate in patients with weakened immune systems or chronic lung conditions.
The research acknowledges a methodological constraint regarding micro-climates. While macro-level warming drives northward migration, localized events create temporary infection corridors. Hospitals have documented surges in Aspergillus loads following dust storms or building renovations. These events fall below the resolution of the current predictive model.
First Appeared on
Source link






