Cancer blood tests are everywhere. Do they really work?
A man in a car looks down at his phone. “No cancer signal detected,” says a message on the screen. The man closes his eyes and smiles in relief.
Why are so many young people getting cancer? What the data say
This television advertisement, aired during American football’s Super Bowl last month, promotes a simple blood test that promises to detect the early signs of more than 50 cancer types — or provide the reassurance of an all-clear.
The test, called Galleri, is one of around 40 such multi-cancer early detection (MCED) tests that are either in development or already on sale. But very few have been through randomized controlled trials (RCTs) — which are considered the gold standard of testing — and none has received approval from regulators.
Last month, the developers of the Galleri test, biotechnology company Grail in Menlo Park, California, released some details from the first RCT of an MCED test. The trial, run in collaboration with the UK National Health Service (NHS), aimed to find out whether Galleri can improve outcomes by reducing the number of cancers detected at advanced stages, when used alongside existing screening programmes — but the results indicate that the test did not meet this goal.
The science that powers these tests is not new: researchers have long known that markers of cancer, including fragments of DNA shed by cancer cells, can show up in blood, saliva and urine. And ‘liquid biopsy’ tests, based on similar technologies to those that underpin MCED tests, are already widely used by physicians to monitor cancer progression and choose the best treatments. Detecting early-stage cancer, however, is much more difficult because of the scarcity of tumour DNA in the blood.
Some researchers say that multi-cancer tests have the potential to revolutionize cancer detection and care, especially for cancer types for which no screening tools are available. Others disagree, highlighting that the tests fail to detect many or even most early cancers, and that the downsides of false diagnoses for many people might outweigh the benefits for a few.
“When you get a negative test, that’s nice, but you could still be harbouring cancer,” says Eric Topol, a physician-researcher and founder of the Scripps Research Translational Institute in San Diego, California. “I think it’s just irresponsible to promote these tests with the data we’ve seen so far.”
Screen time
Around one in five people will develop cancer during their lifetime. It is a leading cause of death in most countries, claiming some 9.7 million lives worldwide in 2022. Detecting the disease in its earlier stages usually means that treatments will be less invasive, cheaper and more likely to result in better survival rates.
Most cancer deaths occur following late-stage diagnoses, when the disease is already advanced and has spread to other parts of the body, says Nitzan Rosenfeld, a cancer biotechnologist at the Barts Cancer Institute, Queen Mary University of London. “That’s because, predominantly, we rely on people noticing something unusual in their body and then going to see their physician,” he says. “The only way around that is to find cancers before they become symptomatic.”
That’s the aim of screening programmes such as mammography for breast cancer; stool testing kits and colonoscopy for colorectal cancer; and smear tests for cervical cancer.
Cheap blood test detects pancreatic cancer before it spreads
These programmes are, however, far from perfect. Some people find the tests painful, embarrassing or inconvenient, so uptake varies. Screening is also unavailable for most cancer types. Preventive screening programmes in the United States detect only 14% of diagnosed cancers — other screening programmes or visits to the doctor do the heavy lifting.
Screening for more types of cancer using MCED tests could increase the chance of people being told they might have cancer when they don’t, potentially causing anxiety and requiring invasive follow-up procedures. These risks are higher for rare cancers, for which it is harder to develop screens.
Although screening has reduced cancer deaths, researchers have, over the past three decades or so, sought fresh approaches that would lower the disease burden further. The potential of MCED tests lies in their simplicity and the promise that they could catch more cancers earlier. “I think they are potentially revolutionary,” says Rosenfeld. “In the best-case scenario, if supported by trial results, they will become a standard of care.”
Clues in the blood
Established screening technologies identify specific cancer types using physiological or molecular indicators, such as signs of changes in breast-tissue density in mammograms. MCEDs have a harder task: identifying biomarkers that are linked to a range of cancers in a single blood sample.
Scientists have long known that cancers spit out bits of themselves — cells and fragments of DNA — into the blood. Many early efforts to develop blood tests for specific cancers examined these fragments for genetic mutations known to be markers of those cancers. But because these cells and fragments from cancers are present only in low concentrations, especially in the early stages, they are masked by the huge amount of other free-floating DNA. For instance, circulating tumour DNA (ctDNA) can make up as little as 0.006% of free-floating DNA in the blood1.
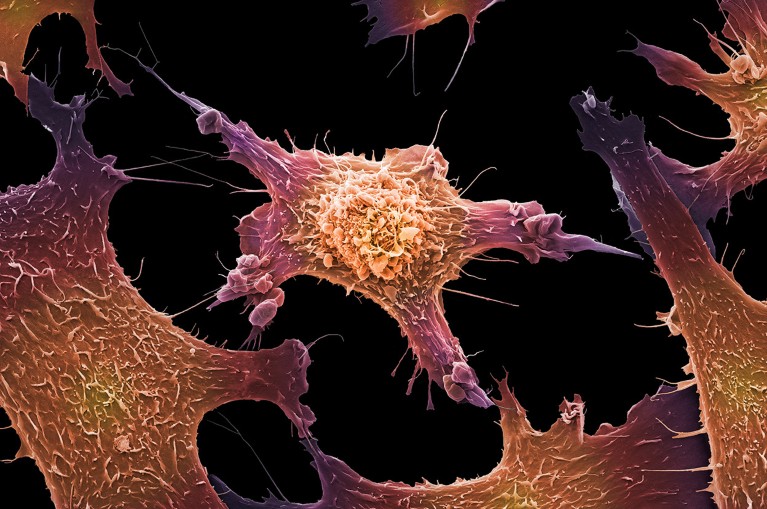
Tumour cells, such as these breast cancer cells, shed fragments of DNA into the bloodstream.Credit: Anne Weston, EM STP, the Francis Crick Institute/Science Photo Library
Improvements in the speed and accuracy of DNA sequencing have gone some way towards overcoming this challenge. In 2008, oncologist Bert Vogelstein at Johns Hopkins University in Baltimore, Maryland, demonstrated that ctDNA could be used to detect the presence of cancer cells and therefore to predict relapse after surgery2.
In 2014, Vogelstein and colleagues went on to show that they could detect ctDNA in more than 75% of people with advanced pancreatic, ovarian, colorectal, bladder, breast and other cancers3, and developed a test called CancerSEEK, which analysed ctDNA alongside cancer proteins and was designed to pick up eight common cancer types. The technology evolved into Cancerguard, an MCED test designed to detect more than 50 types of cancer, which was launched commercially last year by Exact Sciences in Madison, Wisconsin. (In some countries, MCED tests can be marketed without regulatory approval, but if approved they can be more easily covered by insurance and prescribed by physicians.)
Meanwhile, the low concentrations of cancer DNA in blood have led other researchers to take different approaches. “When you’re looking for mutations in plasma, you’re looking for something that is really rare, perhaps one in a million,” says Dennis Lo, a molecular biologist at the Chinese University of Hong Kong.
Lo began assessing DNA fragments in the blood for epigenetic changes: marks left on the genome that turn genes on or off. His team developed a technique that can scan the entire genome for these marks — which are made by a process called DNA methylation — and showed that it could identify the locations of tumours in the body4.
Putting early cancer detection to the test
Grail acquired this technology in 2017 and developed a version that forms the basis of the Galleri test. Many other MCED tests are based on analysing methylation patterns in free-floating DNA in the blood, including PanSeer, developed by Singlera Genomics in San Diego, and OverC, developed by Burning Rock in Irvine, California.
Lo and others have also used the physical appearance of tumour DNA fragments — which differ from regular DNA in various ways, including size and the structure of their ends — to reveal a cancer’s location and type5.
Some research groups and companies developing MCED tests are combining multiple methods to improve accuracy. A team based at the Medical Genetics Institute in Ho Chi Minh City, Vietnam, for example, has developed SPOT-MAS, an MCED test commercially available in southeast Asia, that combines analysis of genetic mutations, methylation status and physical properties of ctDNA to detect liver, breast, colorectal, gastric and lung cancer.
Testing the tests
To assess whether MCEDs are ready for widespread adoption, regulators (such as the US Food and Drug Administration), health-system leaders and insurance providers will look to trials that evaluate tests on a few measures. These include the proportion of people with cancer who have a positive result (sensitivity); the proportion of people without cancer correctly flagged as negative (specificity); and the proportion of people with a positive test result who truly have cancer (positive predictive value). This last measure depends on how common a disease is in the group being tested.
Systematic reviews pooling results from different types of studies have found that MCED tests have high specificity: they can accurately identify those without cancer in 96–99.5% of cases. Their sensitivity is more variable: they correctly catch 30–80% of those with cancer6.
How AI is helping to boost cancer screening
Many MCED tests have been evaluated only in trials in which the participants’ cancer status is already known. These trials might not be representative of wider populations. To deal with this, other studies have instead recruited people with no history of cancer and followed them over time.
One such trial evaluated the CancerSEEK blood test in women aged 65–75 with no history of cancer. It correctly ruled out cancer in 98.9% of people without the disease, or 99.6% when combined with a medical-imaging scan. But it caught only 26 of the 96 cancers diagnosed during the study7.
First Appeared on
Source link