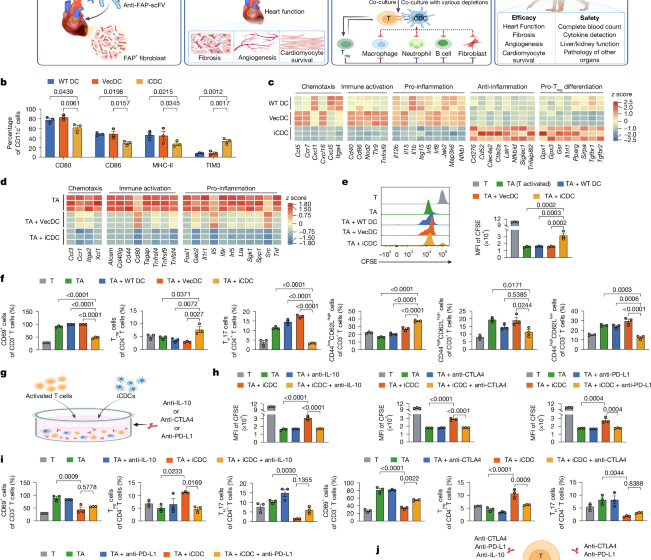
Engineered immunosuppressive dendritic cells protect against cardiac remodelling
Conrad, N. et al. Temporal trends and patterns in heart failure incidence: a population-based study of 4 million individuals. Lancet 391, 572–580 (2018).
Yan, T. et al. Burden, trends, and inequalities of heart failure globally, 1990 to 2019: a secondary analysis based on the global burden of disease 2019 study. J. Am. Heart Assoc. 12, e027852 (2023).
Khan, M. S. et al. Global epidemiology of heart failure. Nat. Rev. Cardiol. 21, 717–734 (2024).
Greene, S. J. et al. Worsening heart failure: nomenclature, epidemiology, and future directions: JACC review topic of the week. J. Am. Coll. Cardiol. 81, 413–424 (2023).
Adamo, L., Rocha-Resende, C., Prabhu, S. D. & Mann, D. L. Reappraising the role of inflammation in heart failure. Nat. Rev. Cardiol. 17, 269–285 (2020).
Markousis-Mavrogenis, G. et al. Immunomodulation and immunopharmacology in heart failure. Nat. Rev. Cardiol. 21, 119–149 (2024).
Sabado, R. L., Balan, S. & Bhardwaj, N. Dendritic cell-based immunotherapy. Cell Res. 27, 74–95 (2017).
Morante-Palacios, O., Fondelli, F., Ballestar, E. & Martinez-Caceres, E. M. Tolerogenic dendritic cells in autoimmunity and inflammatory diseases. Trends Immunol. 42, 59–75 (2021).
Virani, S. S. et al. Heart disease and stroke statistics—2021 update: a report from the American Heart Association. Circulation 143, e254–e743 (2021).
Ambrosy, A. P. et al. The global health and economic burden of hospitalizations for heart failure: lessons learned from hospitalized heart failure registries. J. Am. Coll. Cardiol. 63, 1123–1133 (2014).
Jones, N. R., Roalfe, A. K., Adoki, I., Hobbs, F. D. R. & Taylor, C. J. Survival of patients with chronic heart failure in the community: a systematic review and meta-analysis. Eur. J. Heart Fail. 21, 1306–1325 (2019).
Cohn, J. N., Ferrari, R. & Sharpe, N. Cardiac remodeling—concepts and clinical implications: a consensus paper from an international forum on cardiac remodeling. J. Am. Coll. Cardiol. 35, 569–582 (2000).
Frantz, S., Hundertmark, M. J., Schulz-Menger, J., Bengel, F. M. & Bauersachs, J. Left ventricular remodelling post-myocardial infarction: pathophysiology, imaging, and novel therapies. Eur. Heart J. 43, 2549–2561 (2022).
Mann, D. L. The emerging role of innate immunity in the heart and vascular system: for whom the cell tolls. Circ. Res. 108, 1133–1145 (2011).
Murphy, S. P., Kakkar, R., McCarthy, C. P. & Januzzi, J. L. Jr. Inflammation in heart failure: JACC state-of-the-art review. J. Am. Coll. Cardiol. 75, 1324–1340 (2020).
Monaco, C. et al. Immunotherapy for atherosclerosis. Physiol. Rev. 105, 2141–2230 (2025).
Hackstein, H. & Thomson, A. W. Dendritic cells: emerging pharmacological targets of immunosuppressive drugs. Nat. Rev. Immunol. 4, 24–34 (2004).
Tureci, O. et al. Cascades of transcriptional induction during dendritic cell maturation revealed by genome-wide expression analysis. FASEB J. 17, 836–847 (2003).
Rurik, J. G. et al. CAR T cells produced in vivo to treat cardiac injury. Science 375, 91–96 (2022).
Aghajanian, H. et al. Targeting cardiac fibrosis with engineered T cells. Nature 573, 430–433 (2019).
Du, H. et al. CAR macrophages engineered in vivo for attenuating myocardial ischemia-reperfusion injury. Circ. Res. 137, 846–859 (2025).
Wang, J. et al. CAR-macrophage therapy alleviates myocardial ischemia-reperfusion injury. Circ. Res. 135, 1161–1174 (2024).
Rong, Z. et al. An effective approach to prevent immune rejection of human ESC-derived allografts. Cell Stem Cell 14, 121–130 (2014).
Todorova, D. et al. hESC-derived immune suppressive dendritic cells induce immune tolerance of parental hESC-derived allografts. EBioMedicine 62, 103120 (2020).
Greenwald, R. J., Freeman, G. J. & Sharpe, A. H. The B7 family revisited. Annu. Rev. Immunol. 23, 515–548 (2005).
Sharma, P. et al. Immune checkpoint therapy-current perspectives and future directions. Cell 186, 1652–1669 (2023).
Buchbinder, E. I. & Desai, A. CTLA-4 and PD-1 pathways: similarities, differences, and implications of their inhibition. Am. J. Clin. Oncol. 39, 98–106 (2016).
Balaji, S. et al. Interleukin-10-mediated regenerative postnatal tissue repair is dependent on regulation of hyaluronan metabolism via fibroblast-specific STAT3 signaling. FASEB J. 31, 868–881 (2017).
Saraiva, M., Vieira, P. & O’Garra, A. Biology and therapeutic potential of interleukin-10. J. Exp. Med. 217, e20190418 (2020).
Forte, E., Furtado, M. B. & Rosenthal, N. The interstitium in cardiac repair: role of the immune-stromal cell interplay. Nat. Rev. Cardiol. 15, 601–616 (2018).
Bhattacharya, M. & Ramachandran, P. Immunology of human fibrosis. Nat. Immunol. 24, 1423–1433 (2023).
Kim, J. et al. Wild-type p53 promotes cancer metabolic switch by inducing PUMA-dependent suppression of oxidative phosphorylation. Cancer Cell 35, 191–203, (2019).
Pinto, A. R. et al. Revisiting cardiac cellular composition. Circ. Res. 118, 400–409 (2016).
Yao, L. et al. Spatial multiplexed protein profiling of cardiac ischemia-reperfusion injury. Circ. Res. 133, 86–103 (2023).
Li, Q. et al. Small extracellular vesicles containing miR-486-5p promote angiogenesis after myocardial infarction in mice and nonhuman primates. Sci. Transl. Med. 13, eabb0202 (2021).
Lindsey, M. L. et al. Guidelines for experimental models of myocardial ischemia and infarction. Am. J. Physiol. Heart. Circ. Physiol. 314, H812–H838 (2018).
Lindsey, M. L. et al. Guidelines for in vivo mouse models of myocardial infarction. Am. J. Physiol. Heart. Circ. Physiol. 321, H1056–H1073 (2021).
Xiao, C. et al. FMO2 prevents pathological cardiac hypertrophy by maintaining the ER-mitochondria association through interaction with IP3R2-Grp75-VDAC1. Circulation 151, 1667–1685 (2025).
Hu, X. et al. A large-scale investigation of hypoxia-preconditioned allogeneic mesenchymal stem cells for myocardial repair in nonhuman primates: paracrine activity without remuscularization. Circ. Res. 118, 970–983 (2016).
Li, G. et al. Dynamic molecular atlas of cardiac fibrosis at single-cell resolution shows CD248 in cardiac fibroblasts orchestrates interactions with immune cells. Nat Cardiovasc Res. 4, 380–396 (2025).
Ackers-Johnson, M. et al. A simplified, Langendorff-free method for concomitant isolation of viable cardiac myocytes and nonmyocytes from the adult mouse heart. Circ. Res. 119, 909–920 (2016).
First Appeared on
Source link






