
Forget Memory Loss, Scientists Found a Protein That Reverses Brain Aging, and It’s Already Inside Us
Aging is the one thing nobody escapes, but the creeping decline in brain function has always felt particularly cruel. It happens because our neural stem cells gradually stop doing their job. As neuron production drops, so does our ability to learn and remember. It has long been viewed as a sad but unavoidable part of getting older.
However, a team from the National University of Singapore (NUS) began to wonder if this decline was truly a one-way street. They suspected that if they could understand exactly what goes wrong inside these stem cells, they might find a way to intervene. Their search led them deep into the molecular machinery of the brain, and what they found there offers a genuine glimmer of hope.
The focus of their attention is a protein with the rather clunky name of cyclin D-binding myb-like transcription factor 1, mercifully shortened to DMTF1. This protein acts as a transcription factor, meaning it holds the power to regulate how other genes express themselves.
The Silenced Regulator
When the NUS research team examined aged neural stem cells, they noticed something striking. Using both human stem cell systems and mouse models to simulate the effects of aging, they found that DMTF1 was essentially repressed. It was present, but it had been silenced, leaving the stem cells unable to function properly. According to Derrick Sek Tong Ong, the senior author of the study from NUS published in the journal Science Advances, the mechanisms behind defective neural stem cell regeneration have long remained poorly understood, even though previous studies hinted that the process could be partially restored.
The team then tried an experiment. They restored the expression of DMTF1 in these aged cells. The result was a dramatic turnaround. The stem cells regained their ability to proliferate, effectively getting their regenerative power back. It was a clear indication that this single protein plays a pivotal role in whether neural stem cells thrive or wither as we age.
Bypassing the Telomere Clock
To understand precisely how DMTF1 pulls off this feat, the researchers focused on telomeres. These are the repetitive DNA sequences that cap the ends of chromosomes, protecting them from damage. As an organism ages and cells divide, these telomeres naturally shorten. This shortening is a classic hallmark of aging, as it eventually triggers cellular senescence, which shuts down cell division and promotes inflammation. It has long been considered a biological timer that cannot be reset.
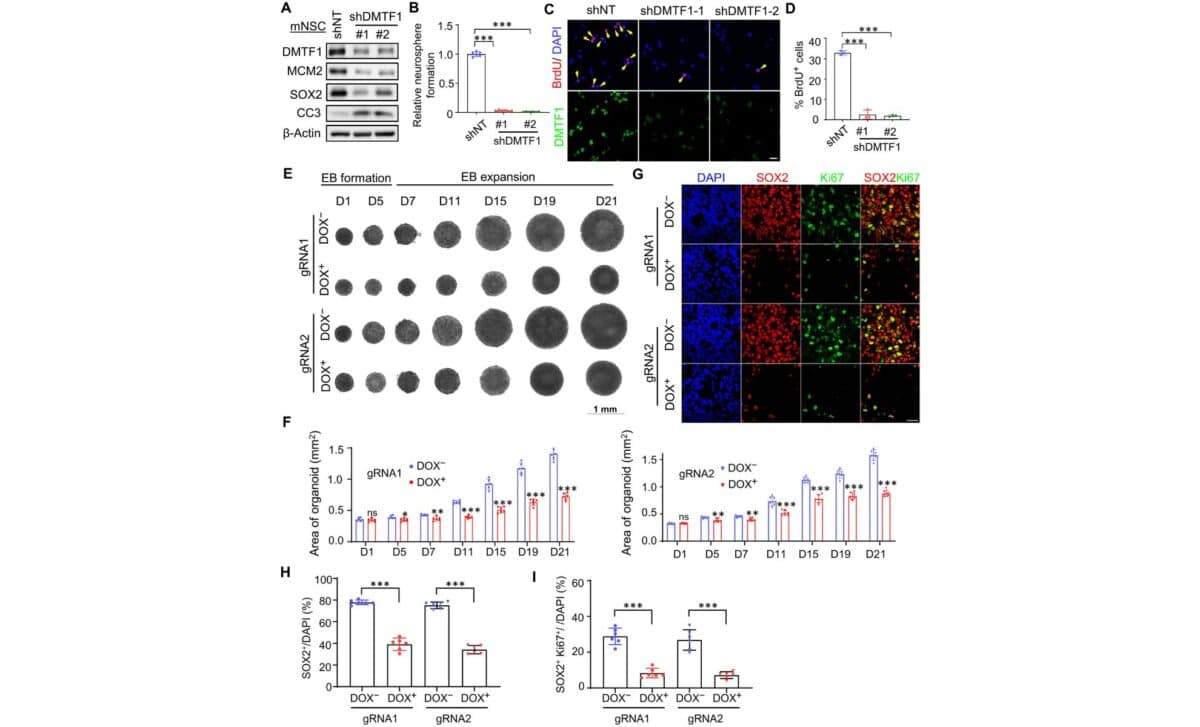
However, DMTF1 appears to cheat the system. Liang Yajing, a co-author of the study from NUS, explained in a press statement that their findings suggest DMTF1 can actively contribute to neural stem cell multiplication even in the context of neurological aging. It achieves this by controlling helper genes that activate growth-related pathways through a process called chromatin remodeling. Crucially, this mechanism rescued the proliferation of stem cells that had already been impaired by telomere shortening, proving that the damage is not necessarily permanent.
A Blueprint for the Future
It is important to manage expectations. All of these experiments were conducted outside of a living human body, meaning we are still in the early days of this research. Liang Yajing herself acknowledged that the study is in its infancy. However, she stressed that the findings provide a vital framework for understanding how the molecular changes associated with aging actually influence neural stem cell behavior. It is one thing to know that stem cells decline; it is another to understand exactly why.
Looking forward, the researchers hope that targeting DMTF1 could eventually lead to therapies that improve neuronal regeneration, even as telomeres inevitably shorten and the aging process continues. As reported by Popular Mechanics, such treatments would not act as a rewind button on aging itself. Instead, they could ensure that the brain maintains its ability to repair and refresh itself, helping to keep those later years genuinely vibrant.
First Appeared on
Source link






