Your Thymus and Your Healthspan
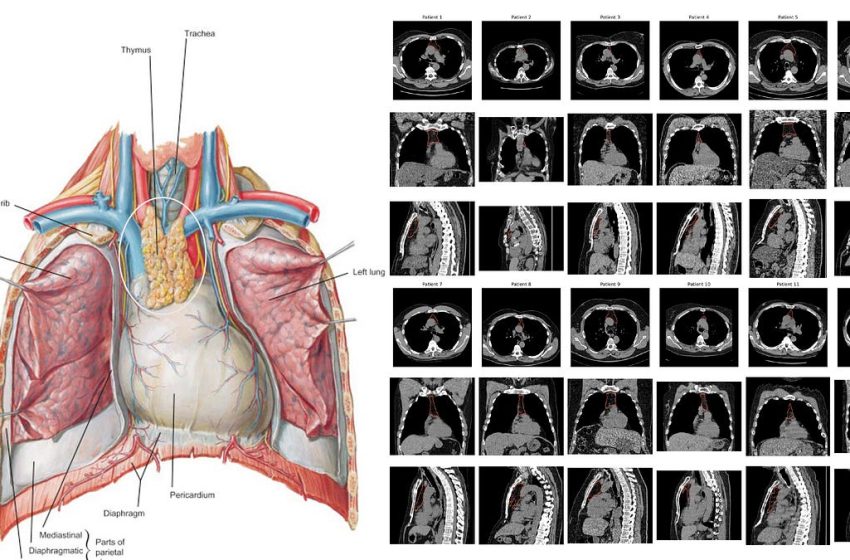
Our thymus gland plays a central role in the development of our immune system, specifically for supporting T cell development (how these cells got their name, maturation and differentiation in the thymus) and discriminating between self and foreign, non-self antigen proteins with production of dendritic cells. As we age, our thymus gland shrinks—the process known as involution—with progressive change from spongy to fatty tissue, with loss of functionality. But this process, with respect to timeline and extent, markedly varies from one person to the next. And to make things even more complicated, our thymus gland anatomy and precise location in the chest also is highly variable. So the famous Frank Netter diagrams from the 1960s (such as at left below) don’t capture the remarkable heterogeneity that a random set of 12 chest CT scans (below at right).
What I consider as 2 landmark papers in Nature this week (here and here) used AI to quantify health of the thymus in 2 large cohorts, and then correlate the metric to a broad array of health outcomes.
In this edition of Ground Truths, I’m going to cover three questions: (1) How was thymus health determined by AI?; (2) How is thymus health linked to key clinical outcomes? and (3) Are there ways we can promote a healthy (aka rejuvenate) thymus gland in our later years?
This was accomplished by extensive work developing and validating an AI pipeline for 2 stages, the first for localization and segmentation of the thymus bed, and the second to quantify a digital marker, termed the thymic health score. There’s a lot to this algorithmic development, so I won’t go into all the details, but just provide a rudimentary outline of what was done. The 1st stage used supervised learning with 2 radiologists reviewing 2,461 chest CT scans. That work trained a 3D U-Net model to automatically identify the thymic bed’s 3D cropped region using center of mass coordinates, which reached 99.8% accuracy.
The 2nd stage used a foundation model that was pre-trained with SwAV (which stands for Swapping Assignment between multiple Views), a self-supervised model, that generated a high-dimensional thymus representation of 4,096 features. That compares to the radiology reductionist and subjective scoring of 0 to 3 where 0 means the thymus is fully degenerated and fatty, and 3 is considered dense, intact glandular soft tissue. Notably, the self-supervised learning (SSL) performance was superior to supervised (AUC ~0.75 vs 0.55, respectively) reflecting the shallowness of the 0-3 classifier vs SSL’s holistic, self-taught AI. The thymic health score ranged from 0 to 100, with the highest number indexed to fully preserved thymic health.
The algorithmic work extended to explainability with both Shapely value distribution, meaning the model was holistic not relying on any “magic” pixel, and occlusion sensitivity, demonstrating the model’s performance was not affected by ribs, sternum, lung tissue, but rather focused on the thymic bed directly.
Below are a few saliency maps from the occlusion sensitivity that indicate the thymic bed focus, not affected by neighboring structures, using the jet color scale.
Now the AI was ready for processing ~25,000 CT scans, each over 30 MB files, from the 2 large participant cohorts to provide a thymic health score, it did so in less than 14 hours, which is less than 2 seconds per scan!
The two cohorts were the National Lung Screening Trial, NLST (N=25,031) and the Framingham Heart Study, FHS (N=2,581) each with baseline demographics and long term follow up of >12 years for health outcomes.
From the NSLT, you can see below that increased age and body-mass index were correlated to reduced thymic health. And, overall, men had reduced thymic health scores compared with women.
There are a lot of graphs in the paper for outcomes, for both the NLST and FHS cohorts. To simplify the major outcomes, below is a composite Figure with striking reduction of all-cause mortality and for the different causes of death. The results were consistent between the 2 cohorts. For example, the cardiovascular mortality hazard ratio average was 0.57 in NLST and 0.38 in FHS (weighted to be 0.53 for the aggregate). The reduction of mortality also extended to digestive diseases and pulmonary disease (data not shown below).
Lung cancer incidence was reduced by 36% for high vs low thymic health (Figure below, with a panel -incidence and b panel-mortality, adjusted for age, sex, BMI, smoking status). Notably, there was a smoker’s paradox: lower incidence and improved survival for lung cancer in smokers who had a high thymus health score.
Smoking was associated with lower thymic scores whereas alcohol was not. Higher thymic health was linked to higher HDL cholesterol, lower triglycerides, lower fasting blood glucose, and lower systolic and diastolic blood pressure. Inflammation biomarkers, such as C-reactive protein and interleukin-6, were elevated in the people with lower thymic health scores.
In their second paper, the same team looked at the relationship between thymic score and outcomes for 3,476 patients receiving cancer immunotherapy. The non-small cell lung cancer progression-free survival was reduced in patients with low thymic health score.
A parallel relationship for survival was seen for other types of cancer including melanoma, breast, and kidney with a 44% lower risk among high thymic scores (Figure). A similar pattern was seen with different immune checkpoint inhibitors.
The thymus health score outperformed programmed death ligand-1 (PD-L1)and tumor mutation burden (TMB) assay of the tumor and was an independent prognostic marker of progression-free survival. In this dataset, a direct connect with thymus score and adaptive immune function was noted with a correlation before cancer treatment for both T-cell diversity and thymus T-cell production (a metric known as T cell receptor excision circles).
Thymus Removal Study
There is a highly relevant citation about thymus function before moving onto ways to rejuvenate it. In 2023, a very important study of thymus removal during cardiothoracic surgery was published, with 1146 patients undergoing thymus removal vs 1146 matched controls (mean age 55 years). The adverse outcomes for thymus removal were striking, with a 2.9 higher risk of all-cause mortality and doubling of cancer incidence, 1.5 fold increase in cancer mortality, and 1.5-fold increase in autoimmune diseases. The immune function showed marked compromise after thymus removal, as reflected by CD4+ and CD8+ T cells (Figure below)
In recent months we’ve learned a lot about the process of thymus involution and the pathways by which this may be modulated. Thymus involution is primarily due to loss to thymus epithelial cells (TECs) and there are 2 subtypes with different functions: the cortical, responsible for positive selection of T cells, and the medullary, for negative selection. When adipose tissue infiltrates the thymus, thymic adipocytes are pro-inflammatory, knocking out T-cell output. Likewise, systemic inflammation, or “inflammaging,” from smoking, obesity, and chronic stress promote thymic adipocytes. Now, from preclinical studies, we know about key pathways that account for these processes.
Historically, back in 2014, FOXN1 was recognized as a single master transcription factor essential for TEC function, with studies in mice with forced FOXN1 upregulation leading to thymus regeneration. That laid the foundation for potential regrowth of quiescent thymus cells. It took awhile to find pathways to accomplish this goal and to even refute its role as the master regulator.
Feng Zhang and his team recently showed that injection of an mRNA vaccine to the liver encoding DLL1, IL-7 and FLT3-L in combination, factors known to support thymopoiesis (Figure), successfully enhanced immune function in aged mice. However, the effect was transient.
The FGF21 Story
Two reports in 2025 addressed the thymus gland’s production of FGF21, a growth factor (fibroblast growth factor 21) and its control of thymus involution. This growth factor is also produced by the liver, but it doesn’t have impact on the thymus in aged rodent models. In contrast, The liver’s hepatocyte growth factor (HGF) has been shown to reverse senescent TEC structural changes. Growth hormone stimulates production of insulin growth factor-1 (IGF-1) in the liver and the thymus. Two small studies in human participants (TRIIM, for Thymus Regeneration, Immunorestoration, and Insulin Mitigation) of 6 and 50 people, respectively, treated with combined human growth hormone, metformin and DHEA, suggested the potential of slowing epigenetic aging and improving thymus mass and function. But the combination of drugs, small sample, and lack of controls make conclusions murky.
Increased thymus FGF21 led to increased CD8 T cells in old mice and extended healthspan and improved physical performance. Building on the previous FOXN1 data, ablation of β-klotho, the obligatory co-receptor for FOXN1, accelerated thymic aging. The benefit to FGF21 was confirmed in a companion paper using an FGF21 knock-in mouse model showing thymus enlargement and increased TECs throughout the lifespan. (schematic Figure below). The accompanying editorial for these 2 papers speculated: “The proposed link between age-associated thymus involution and organismal aging is intriguing, and suggests we might seek to achieve systemic rejuvenation by counteracting thymic involution.”
Lessons from the Axolotl (Mexican Salamander)
In an elegant set of experiments of the axolotl (Figure), known for its complete thymus regeneration after total surgical removal, mediators were identified. Surprisingly FOXN1 was dispensable. But midkine, a growth factor, appeared to be the driver, the initiator of thymus cell growth, survival, and repair. Other components that contributed were Postn+ (periostin-expressing mesenchymal niches) and Ccl19, a chemokine protein from the bloodstream.
RANK and RANKL
RANK (receptor activator of nuclear factor κB its ligand (RANKL) have been considered key regulators of medullary TECs. In a study of both aged mice and human thymus cell cultures, their key role was confirmed, with multiple benefits of resorting TEC function, recruitment of progenitor cells, and T cell development.
This body of work strongly supports the health of the thymus gland as a critical regulator of human healthspan, not just a correlate or link. The new landmark studies are reinforced by the regenerative biology experimental models that have shown, via an array of mediators, that healthspan of aged mice is at least in part dependent on thymus gland function and its critical role for maintaining adaptive immunity. It sure seems that we’d be better off having an immune reservoir of naive T cells instead of a profile in older adults of exhausted, low quality, memory T cells, with a senescent phenotype.
It is striking that we ignored the importance of the thymus gland for many decades. A wake-up call was the results of the thymectomy matched control study reviewed here, with the opposite of high thymic health score improved health outcomes.
The convergence of AI and healthspan here is notable. The exceptional and laborious work for developing and validating (no less explaining) the AI to quantify thymus health set the foundation for probing the connect the major health outcomes in two cohorts with extensive follow-up. We’ve never had a way to meaningfully quantify the thymus gland before, so this represents important leverage of supervised, self-supervised, and transformer models. The study wouldn’t have been possible without AI. And it begs the question as to whether there are ways we can maintain our thymus at a high health score.
But it’s not so simple to rejuvenate our thymus. There are multiple risks including the induction of autoimmunity, increasing the risk of cancer, and inducing a pro-inflammatory state. For example, factors that increase TEC proliferation could compromise the medulllary negative selection process and leak out auto-reactive (self-attacking) T cells. Thymic cells could be induced to proliferate, but we know, for example, that growth hormone and IGF-1 are associated with an increase risk of cancer. If thymus rejuvenation doesn’t clear out the accumulated senescent cells in older adults, then inflammation within the gland could block the production of TECs. Keeping these risks in mind, we now have identified many ways to keep our thymus healthy as we age that undoubtedly will be tested in clinical trials going forward. Ultimately, the benefit-to-risk tradeoffs will be defined.
In the meantime, we have desperately needed an immunome. As I’ve stressed in multiple prior Ground Truths, we have no test in the clinic to assess a patient’s immune status! It was very encouraging for me to get an email from Hugo Aerts, the senior author of the landmark papers in the days after they were published: “I also wanted to say that your Substack on “Why We Need an Immunome” was inspiring for this work. We found it a very compelling perspective and greatly enjoyed reading it.”
Just having an assessment of a person’s immune status via a chest CT scan, with millions of these performed per year, might help in guiding the right immunotherapy for cancer (e.g. more intensive, combinations for those with low thymic health scores) and help define people who are increased risk for age-related diseases of cancer, cardiovascular, and neurodegenerative. From a practical standpoint, with 20 million chest CT scans performed each year in the United States, opportunistic AI-derived thymic health scores could be routinely provided as part of their interpretation.
The main theme of Super Agers is that we now have many layers of data (genetics, proteins, biomarkers, organ clocks that can be integrated via multimodal AI) for us to define a person’s specific risk decades before one of these age-related diseases leads to symptoms, enabling prevention. From the new studies this week, we can add thymus health score as yet another way to understand a person’s risk, a window into their adaptive immune system and healthspan. And perhaps someday we will be able to safely rejuvenate the thymus, or maintain its health through one’s life, and use the thymus health score to monitor.
NB: I wrote this post (No A.I.)
A Quick Poll
*********************************************************************
Thanks to Ground Truths subscribers (now > 200,000) from every US state and 212 countries. Your subscription to these free essays and podcasts makes my work in putting them together worthwhile. Please join!
If you found this interesting PLEASE share it!
Paid subscriptions are voluntary and all proceeds from them go to support Scripps Research. They do allow for posting comments and questions, which I do my best to respond to. Please don’t hesitate to post comments and give me feedback. Let me know topics that you would like to see covered.
Many thanks to those who have contributed—they have greatly helped fund our summer internship programs for the past two years. It enabled us to accept and support 47 summer interns in 2025! We aim to accept even more of the several thousand who will apply for summer 2026.
First Appeared on
Source link


















